Research
- The Hovestadt Lab studies pediatric brain tumors, one of the leading causes of death in children
- We develop and apply modern genomic and computational tools to improve our understanding of these tumors
- A main goal of our research is to translate our findings into the clinic to improve patient care
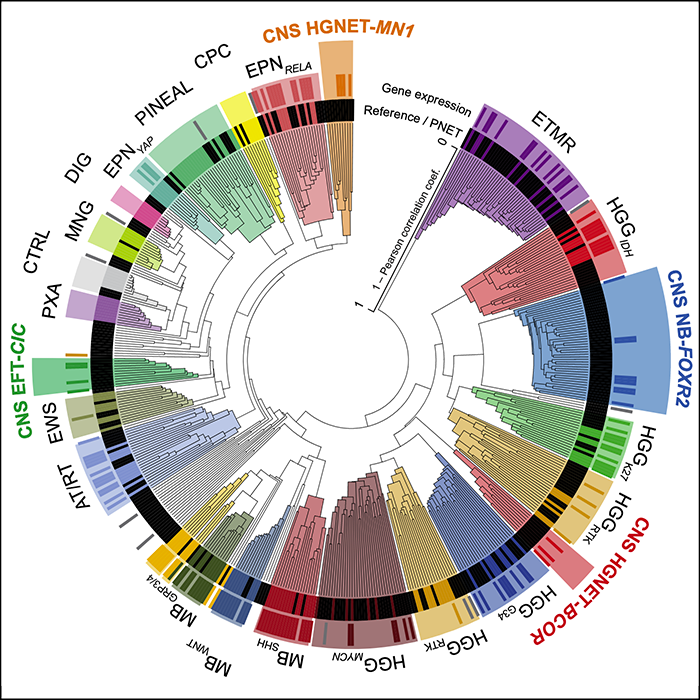
There are many types of brain tumors, and the list is growing
The brain is the most complex organ of the human body. It consists of a vast number of distinct cell types that vary greatly in abundance throughout development. This is paralleled by a large number of different types of brain tumors that occur in children and adults. We have established genome-wide DNA methylation profiling as an effective tool for classifying these tumors. DNA methylation is a central epigenetic modification, and its biological and physical properties make it an ideal biomarker for distinguishing different tumor types in an unbiased manner using computational algorithms. As as result, the profiling of increasingly large patient cohorts has enabled the discovery of increasingly rare tumor types. Their molecular definition represents a first step to enable targeted biological research and more accurate outcome predictions and treatment decisions in the clinic.
Learn more: Sturm et al., Cancer Cell 2012 • Hovestadt et al., Acta Neuropath 2013 • Sturm et al., Cell 2016 • Capper et al., Nature 2018
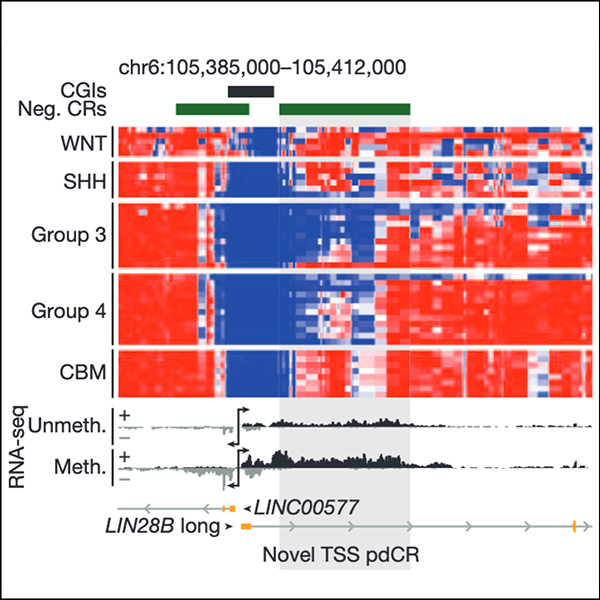
Some pediatric brain tumors are not caused by genetic mutations
Cancer is often described as a genetic disease, in which somatic gene mutations cause uncontrolled cell proliferation. Interestingly, most pediatric cancers contain much fewer mutations than adult cancers. Some pediatric brain tumors even do not harbor any mutations in known cancer genes, raising the important question of why these tumors occur. We believe the answer lies in how genes are regulated through epigenetic modifications. In fact, many genes in the human genome that fulfill specific roles during normal development can become oncogenes if activated in the wrong cellular context. One of the goals of the lab is to uncover cancer-causing epigenetic variants and to use this information to better understand tumor-driving mechanisms and inform novel treatments.
Learn more: Hovestadt et al., Nature 2014
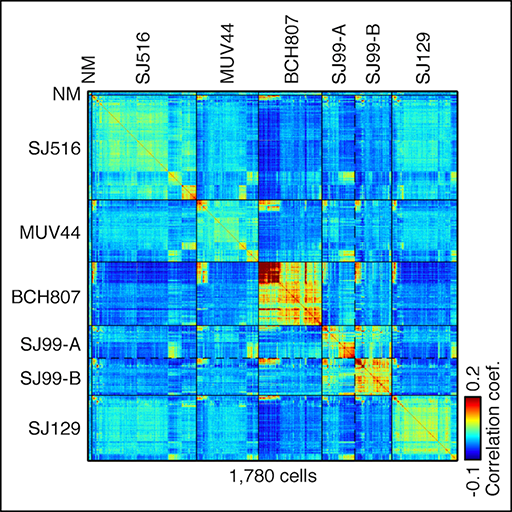
All tumor cells originate from the same cell-of-origin, but they are not equal
We are very interested in understanding how individual cells differ within the same tumor. Cells can differ genetically, reflecting hierarchies of tumor subclones that arise through clonal evolution over time. Cells can also differ functionally, reflecting developmental trajectories of malignant cells such as cancer stem cells and their more differentiated progeny. All these cells compete and cooperate within the cancer ecosystem. We develop and apply single-cell genomic techniques to characterize these ecosystems and find new ways of targeting dependencies between malignant cell types and with cells of the microenvironment. We are also very interested in understanding the early steps of tumorigenesis and how patient tumors evolve over time to adapt to different treatments.
Learn more: Filbin et al., Science 2018 • Hovestadt et al., Nature 2019 • van Galen et al., Cell 2019
Diagnosing brain tumors is difficult – computers can help
Brain tumors are commonly diagnosed based on their histopathological appearance and an increasing number of select single-gene/locus molecular tests. Even for experienced neuropathologists this can be challenging, as there are over 100 different types of brain tumors that vary greatly in their rate of occurrence, leading to significant inter-observer variability. We believe that cancer diagnostics is a task predestined for machine learning algorithms. Using genome-wide DNA methylation profiles of clinical samples as a molecular marker, we created a machine learning-based classification system that produces an accurate and reproducible diagnosis. This system has become routine clinical assay that has been applied in tens-of-thousands of brain tumor patients around the world, likely improving treatment decisions in many cases.
Learn more: Capper et al., Nature 2018
